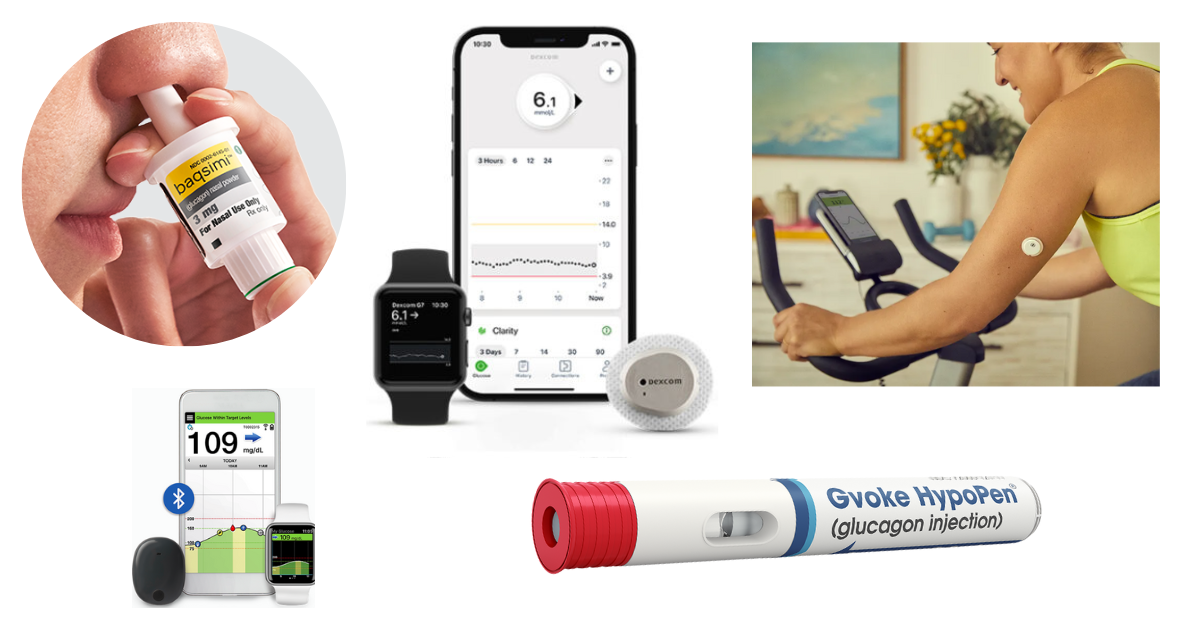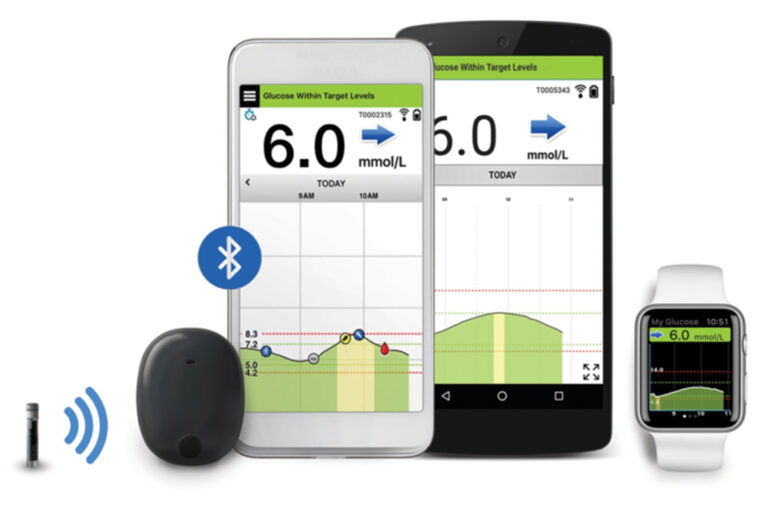
Eversense E3 CGM Approved for Two Sensors per Year: Your “Happily Ever(sense) After” - Taking Control Of Your Diabetes®
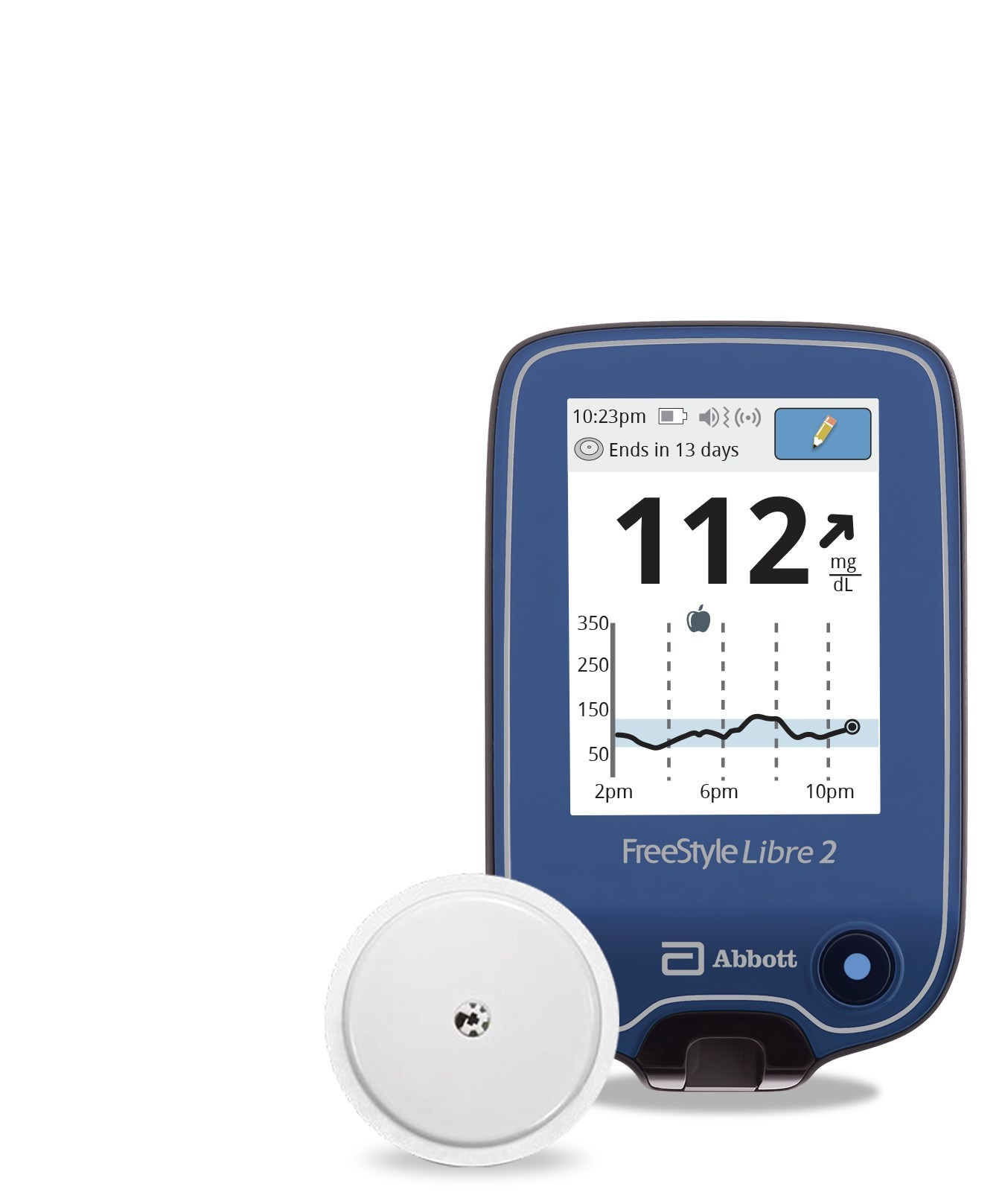
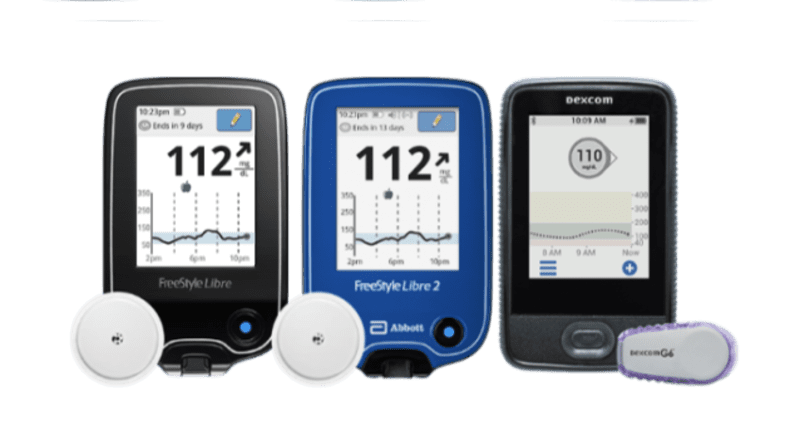
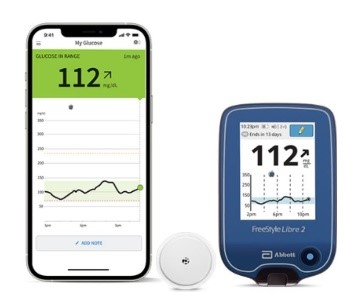
Abbott's FreeStyle® Libre 2 iCGM Cleared in U.S. for Adults and Children with Diabetes, Achieving Highest Level of Accuracy and Performance Standards - Jun 15, 2020

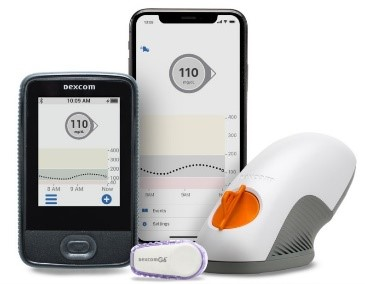
OneTouch Vibe Plus Insulin Pump Earns FDA Approval And Health Canada License And Is First Pump Integrated With The Dexcom G5 Mobile Continuous Glucose Monitor

For People With Type 1 Diabetes, An 'Artificial Pancreas' Is Almost Here : Shots - Health News : NPR





:max_bytes(150000):strip_icc()/Eversense-CGM-768x499-d920f1a66ddc42729d274ec509603bf6.jpeg)








![Freestyle Libre 3 - [Pain-Free Blood] Glucose Monitoring - 2023 Freestyle Libre 3 - [Pain-Free Blood] Glucose Monitoring - 2023](https://cgmmonitors.com/wp-content/uploads/2023/01/All-You-Need-to-Know-About-Freestyle-Libre-3.jpg)