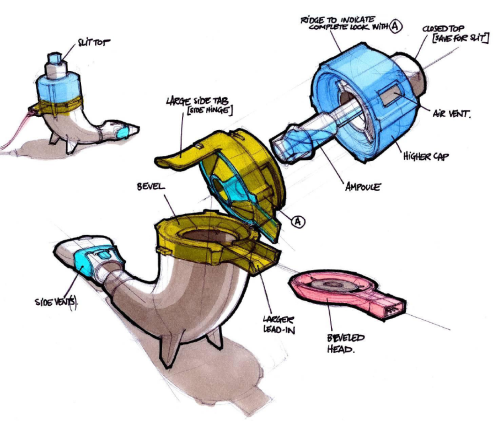
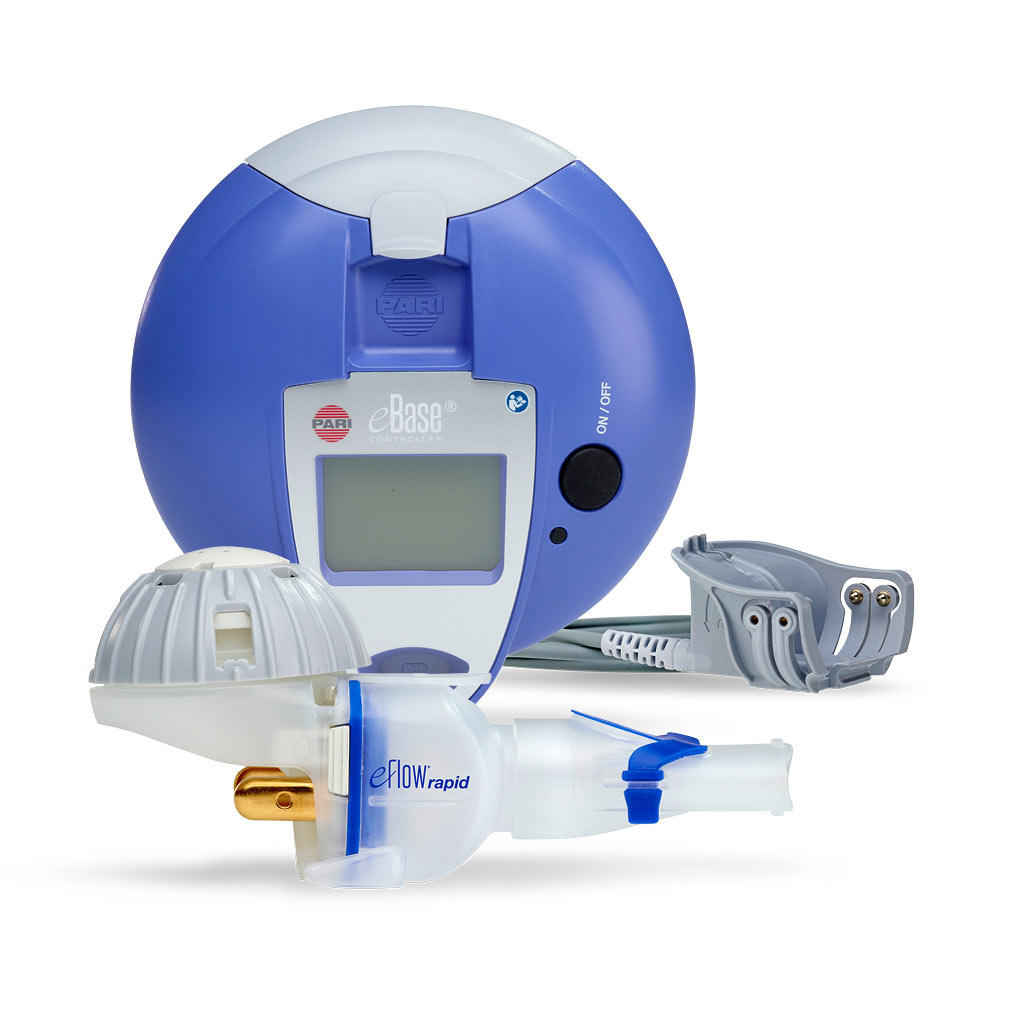
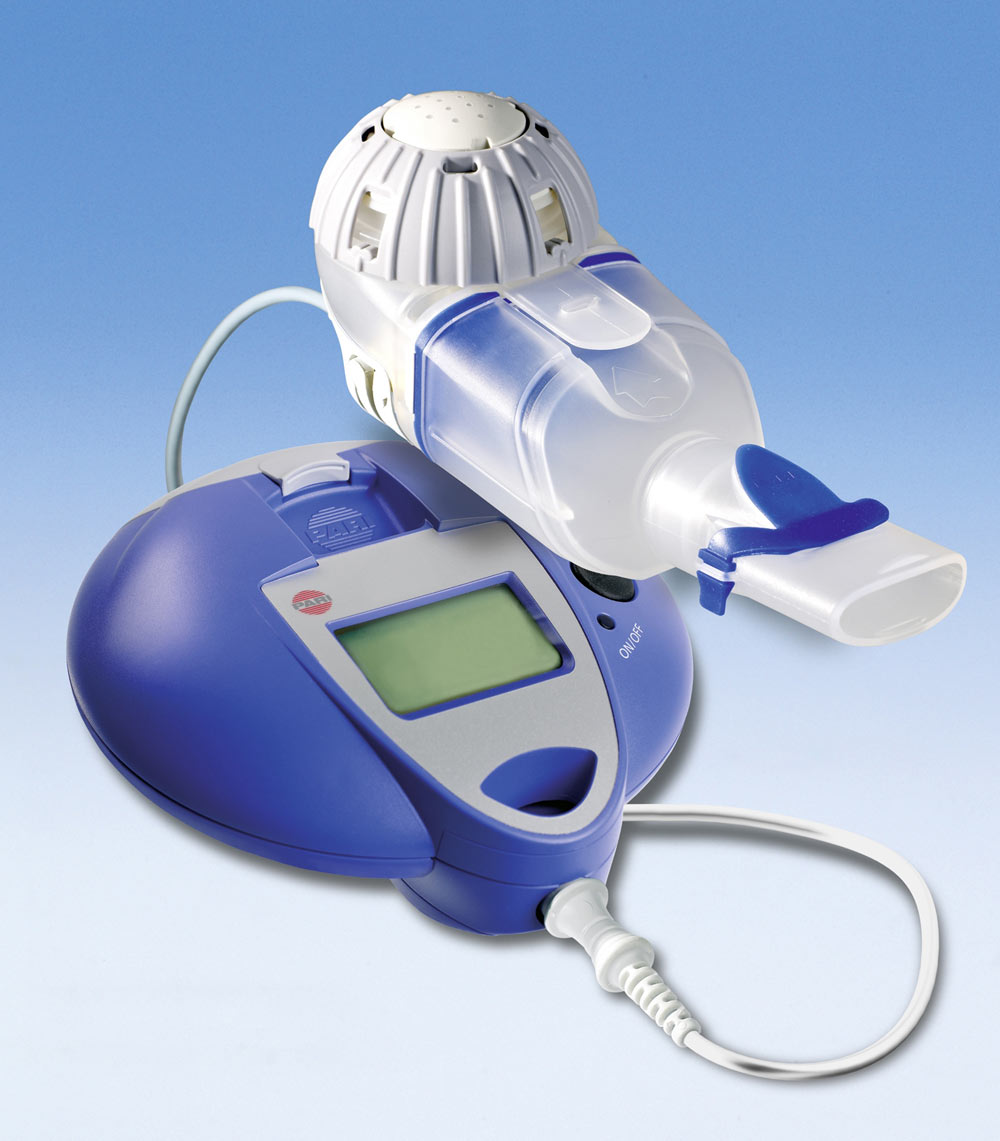
eFlow Ò Closed System main components: handset unit, controller, and... | Download Scientific Diagram
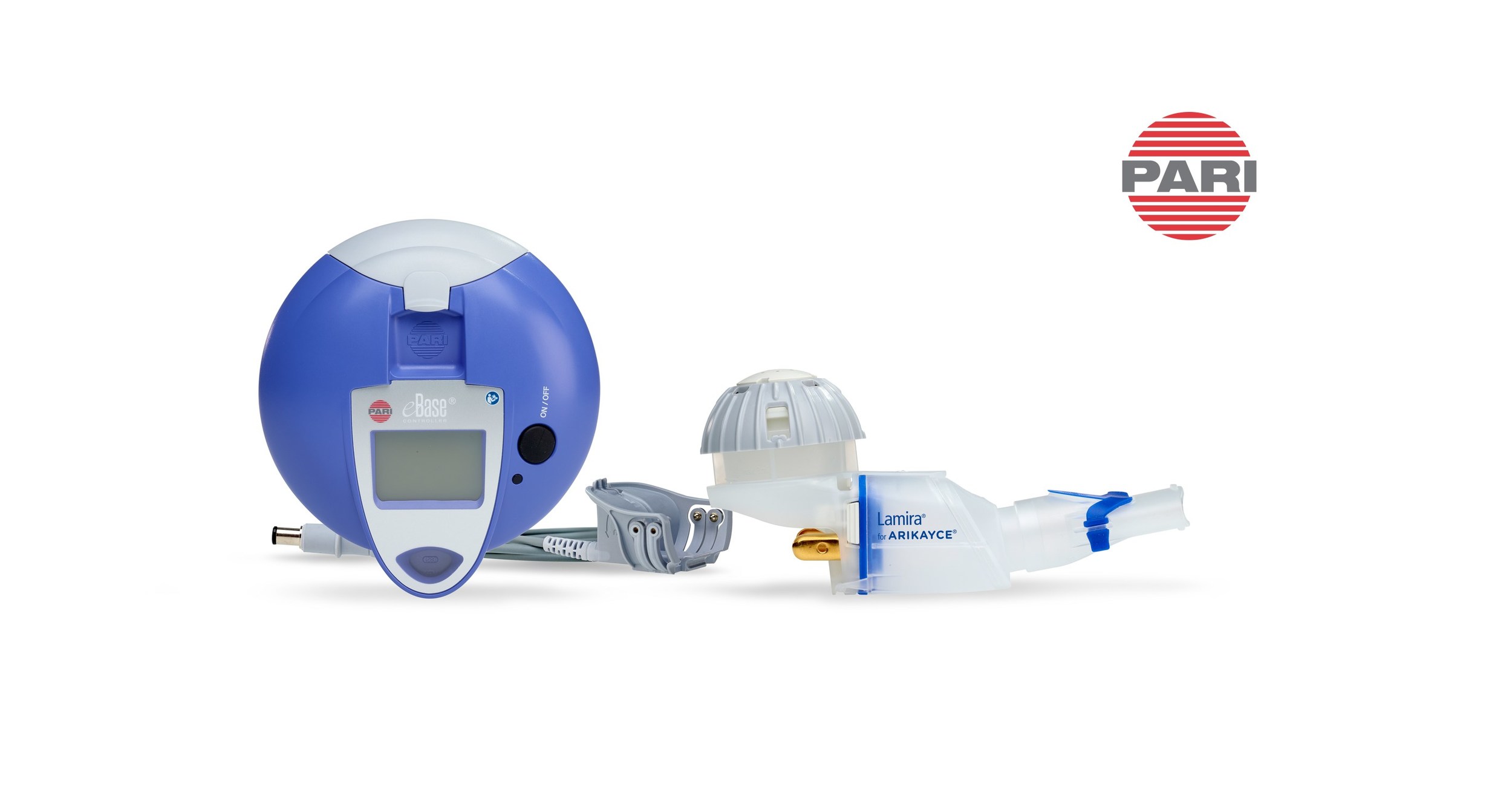
LAMIRA®, an eFlow® Technology nebulizer, expected to launch in Japan as a medical device for exclusive administration of ARIKAYCE® (amikacin liposome inhalation suspension)
Impact of baseline clinical features on outcomes of nebulized glycopyrrolate therapy in COPD | npj Primary Care Respiratory Medicine
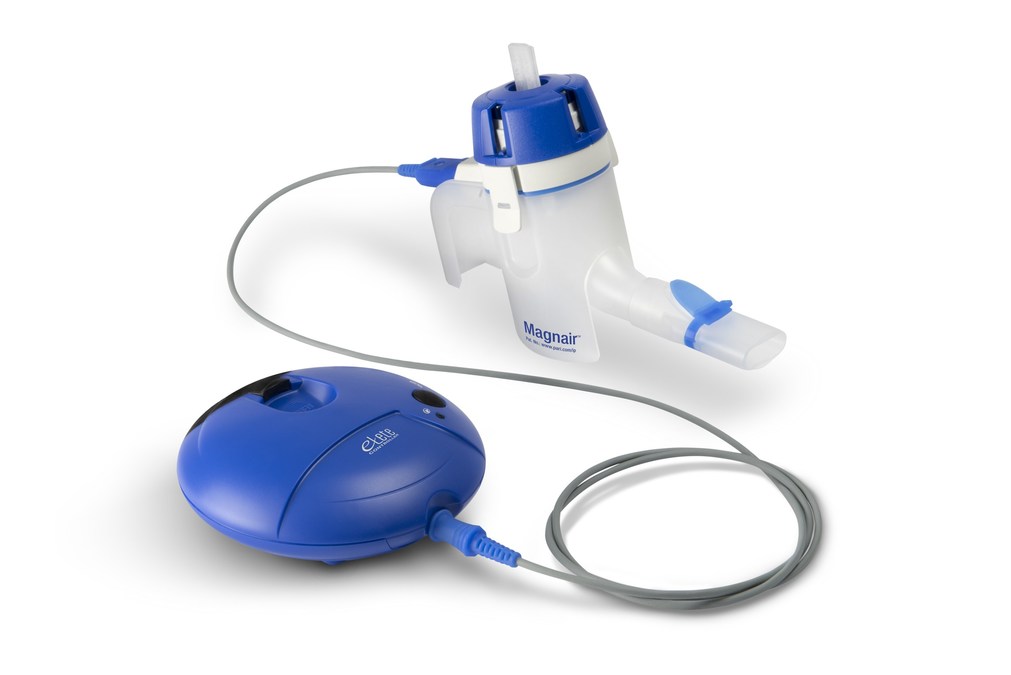
Magnair™ an eFlow® Closed System Nebulizer together with Sunovion's Lonhala™ is the first eFlow technology based product to receive FDA Approval to Treat Chronic Obstructive Pulmonary Disease (COPD)

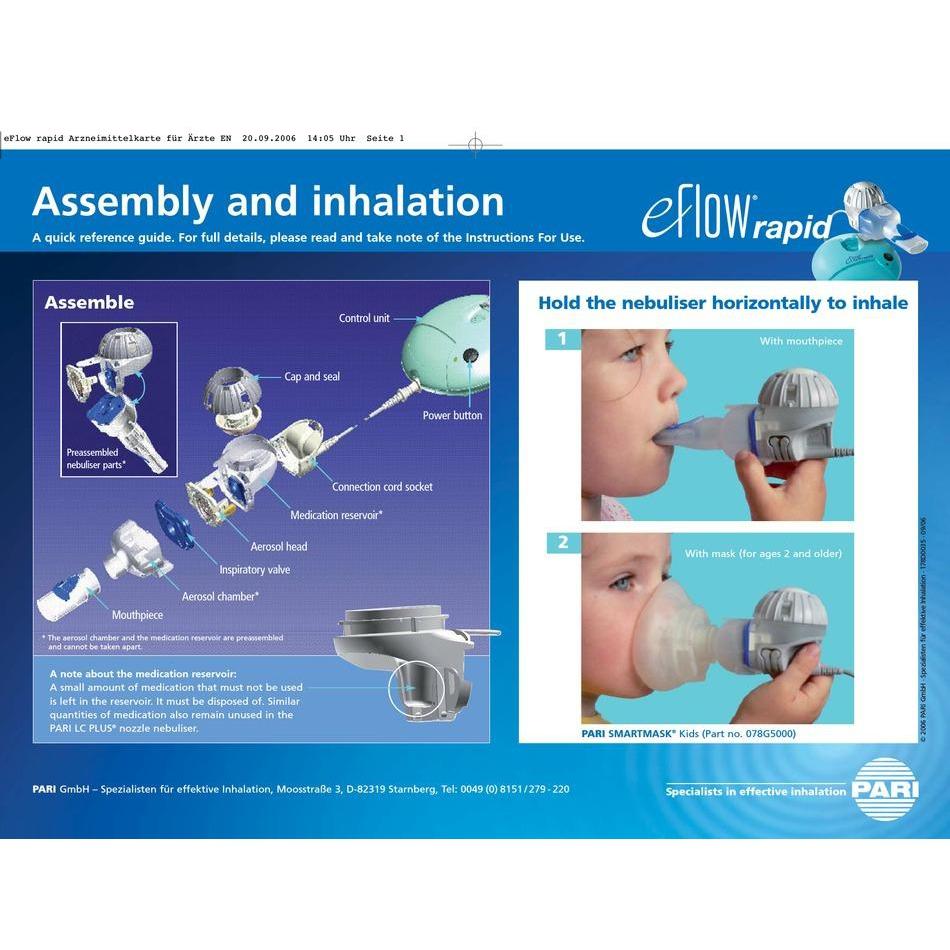
A schematic of the aerosol production within the eFlow® nebulizer system | Download Scientific Diagram

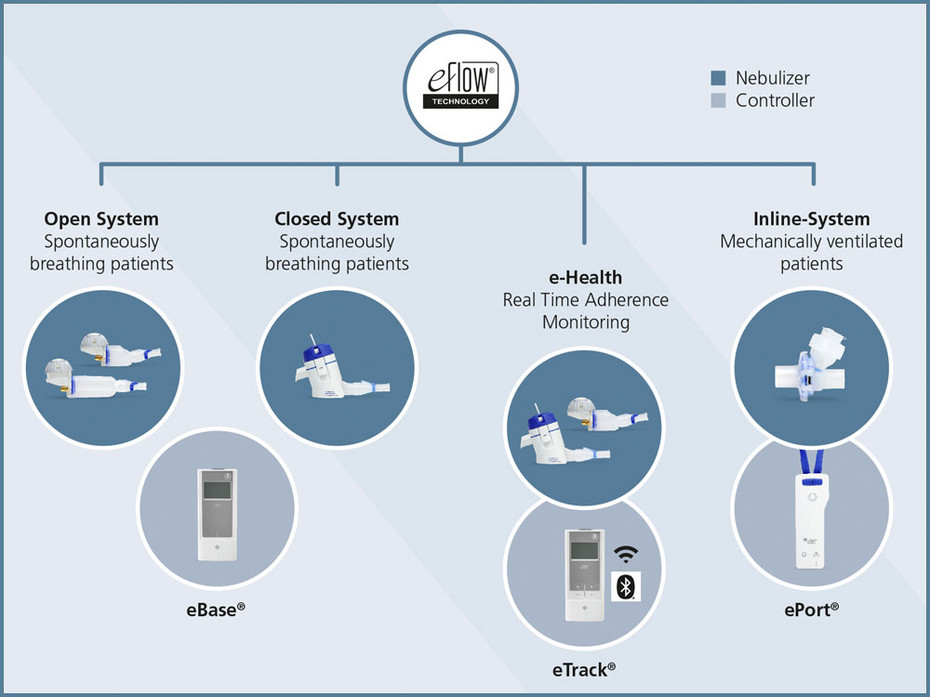
A configuration of the eFlow platform, showing components that can be... | Download Scientific Diagram

In Vitro Characterization of the eFlow Closed System Nebulizer with Glycopyrrolate Inhalation Solution | Journal of Aerosol Medicine and Pulmonary Drug Delivery
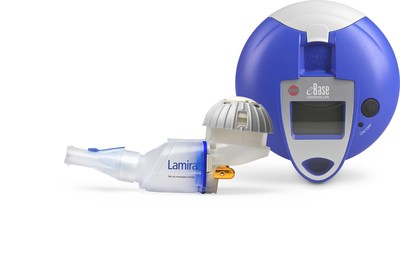
PARI Pharma's eFlow® Technology device, LAMIRA™, approved as the only nebulizer system to deliver Insmed's ARIKAYCE® (amikacin liposome inhalation suspension) | Markets Insider